AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Carbon molar mass grams12/5/2023 The molecular mass of carbon dioxide CO 2 is 44 g/mole. Molecular mass of carbon dioxide ( CO 2) ( Atomic mass of carbon C) + ( Atomic mass of Oxygen) x 2. The mass number is nothing but the sum of the number of protons and number of neutrons present in the nucleus of the atom. The chemical formula of Carbon dioxide is CO 2. Molecular weight is also called mass number. Therefore, the answer is – option (c) – Gram molecular mass of carbon is 12g It has an atomic mass of 12 atomic mass units or 12g. It is a p-block element, present in Group 14 and 2nd period. To calculate the mass of a single atom of carbon, we just need to divide the molar mass of 12.0 g (0,012 kg) by the number of particles per mole (Avogadros. \Ĭarbon is an element with atomic number 6. Gram molecular mass of carbon is: 6g 11g 12g 14g Gram molecular mass is the mass in grams of one mole of a molecular substance. 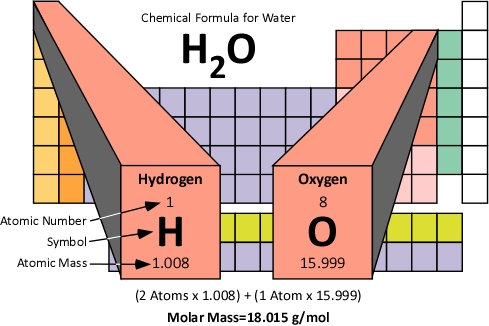
Molar mass of an element and atomic mass of an element have the same value. It is due to the fact that the atoms of different elements contain different numbers of protons, neutrons, and electrons, and therefore they have different masses. All elements have different molar masses. equal to 6. It should be noted that each element has its own unique molar mass. Avogadros number (mole) - number of atoms in 12g of pure carbon-12. “The mass in grams of one mole of substance is called molar mass”. Step 2: Mass of one atom of carbon - 12: Mass of one mole of carbon - 12 12 g. Step 1: Molar mass of carbon ( C): Molar mass of carbon 12 g mol - 1. The elementary entities may be an atom, a molecule, an ion, an electron, or any other particle.  
Refer to the periodic table to solve this question. This number is called Avogadros constant. Atomic number refers to the number of electrons present in the element. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6. Hint: Do not confuse atomic mass with atomic number. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
